Telix Full Year Results 2025 Investor Webcast Notification
Telix advises that it will release its full year results for the period ended 31 December 2025 on Friday 20 February 2026 AEDT (Thursday 19 February 2026…
Read more


News & Views
Telix advises that it will release its full year results for the period ended 31 December 2025 on Friday 20 February 2026 AEDT (Thursday 19 February 2026…
Read more
Telix today announces that the first patient has been dosed in the Phase 3 registration study of TLX591-Px (Illuccix®, Kit for the preparation of 68Ga-PSMA-11) for prostate cancer imaging in…
Read more
Telix provides an update on its commercial and operational performance for the quarter ended 31 December 2025 (Q4…
Read more
News,
Telix today announces that the Chinese National Medical Products Administration (NMPA) Center for Drug Evaluation (CDE) has accepted the filing of a New Drug Application (NDA) for TLX591-Px (Illuccix®, Kit…
Read more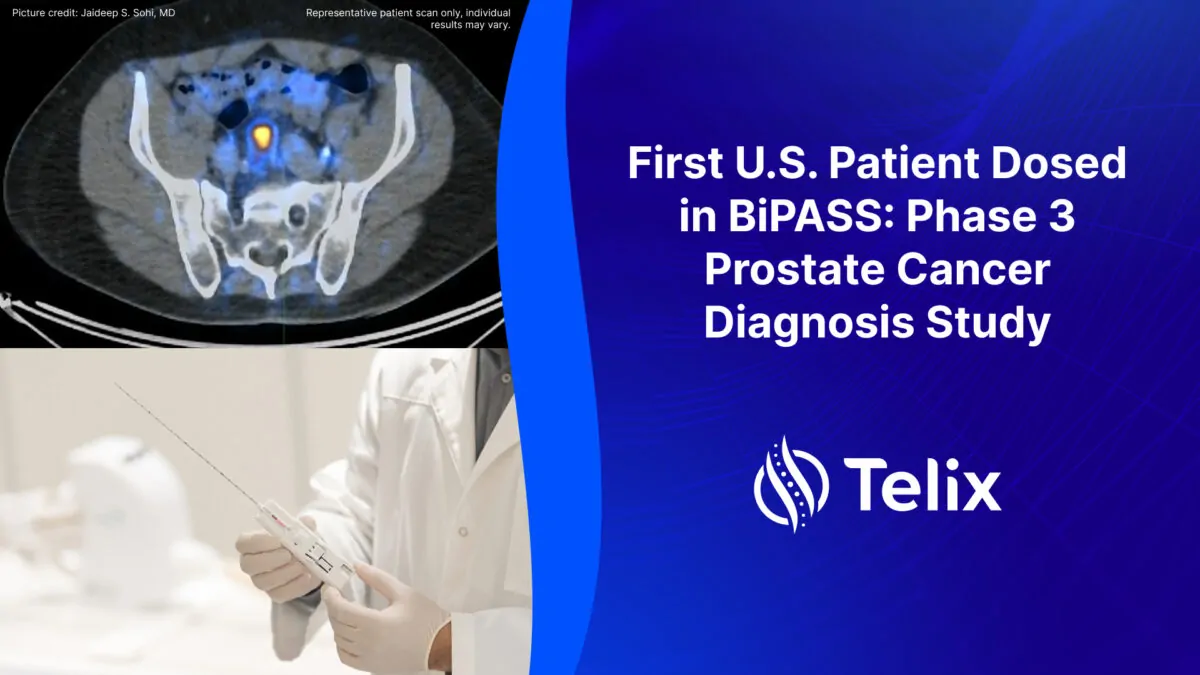
Telix today announces that the first patient in the United States (U.S.) has been dosed in BiPASS™ (Biopsy of the Prostate Avoidance Stratification Study), Phase 3 trial to evaluate the…
Read more
Telix is pleased to provide a copy of the presentation that will be delivered by Dr. Christian Behrenbruch, Managing Director and Group CEO at the 44th Annual J.P. Morgan Healthcare Conference…
Read more
Telix today announces that final results from the ZIRDOSE-CP Phase 1 study of TLX250-CDx (Zircaix®, 89Zr-girentuximab) PET imaging in Chinese patients with suspected clear cell renal cell carcinoma (ccRCC) have been published…
Read more
Telix today advises that Dr. Christian Behrenbruch, Managing Director and Group CEO, will be presenting at the 44th Annual J.P. Morgan Healthcare Conference being held in San Francisco, CA…
Read more
ASX,
Telix today provides a precision medicine portfolio update in relation…
Read more
Telix today provides an update to confirm the current status of the ProstACT Global Phase 3 study of its lead prostate cancer therapy candidate TLX591 (lutetium (177Lu) rosopatamab tetraxetan) in…
Read more