First Patient Dosed in Study of Targeted Alpha Therapy Candidate for Bladder Cancer
Telix announces that a first patient has been dosed in the ‘PERTINENCE’ Phase I study of TLX250-CDx in patients with non-muscle-invasive bladder...
Read more


Latest News
Category: Clinical
Telix announces that a first patient has been dosed in the ‘PERTINENCE’ Phase I study of TLX250-CDx in patients with non-muscle-invasive bladder...
Read more
Telix is pleased to announce that the first Australian patient has been dosed in the international NOBLE Registry, a global study that aims to improve access for men to state-of-the-art...
Read more
Telix is pleased to announce that a first patient has been dosed in a Phase II study of TLX250-CDx (89Zr-DFO-girentuximab) in patients with triple-negative breast cancer (TNBC) at the Institut...
Read more
Telix welcomes the Society of Nuclear Medicine and Molecular Imaging (SNMMI) updated Appropriate Use Criteria (AUC) for prostate specific membrane antigen (PSMA) positron emission tomography (PET)...
Read more
Telix welcomes the updated NCCN Guidelines® for prostate cancer, which includes PSMA PET imaging modalities, including Ga-68...
Read more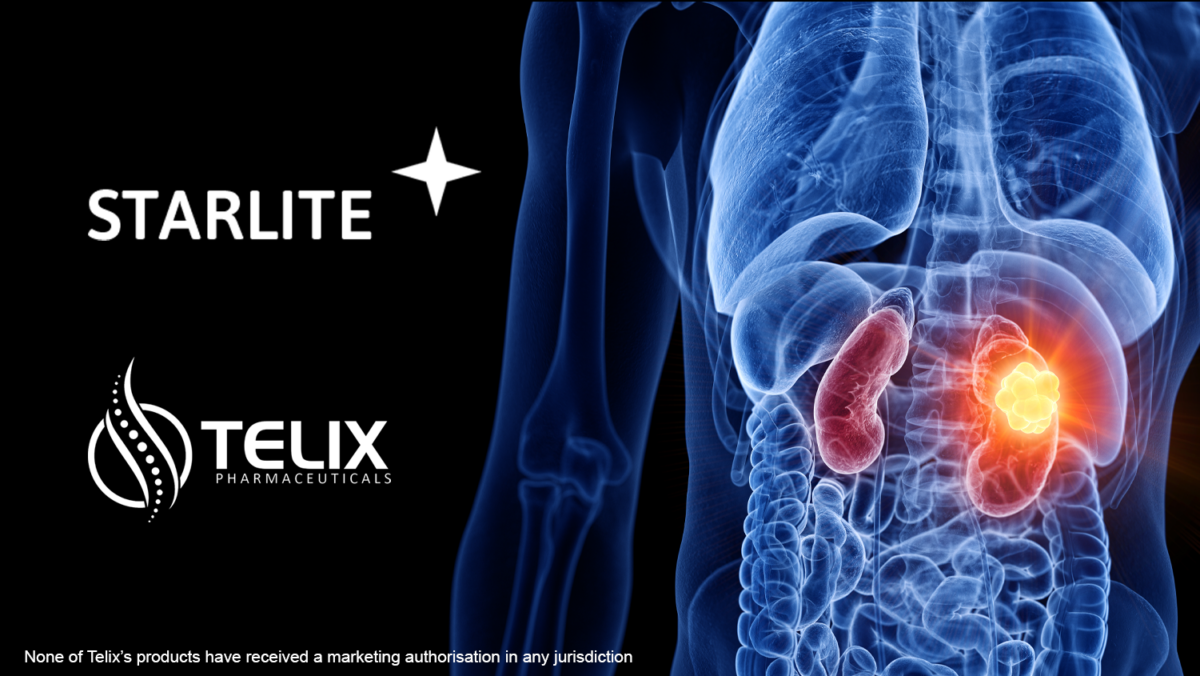
Telix announces the FDA has accepted the IND Application to undertake a clinical study of Telix’s investigational kidney cancer therapy,...
Read more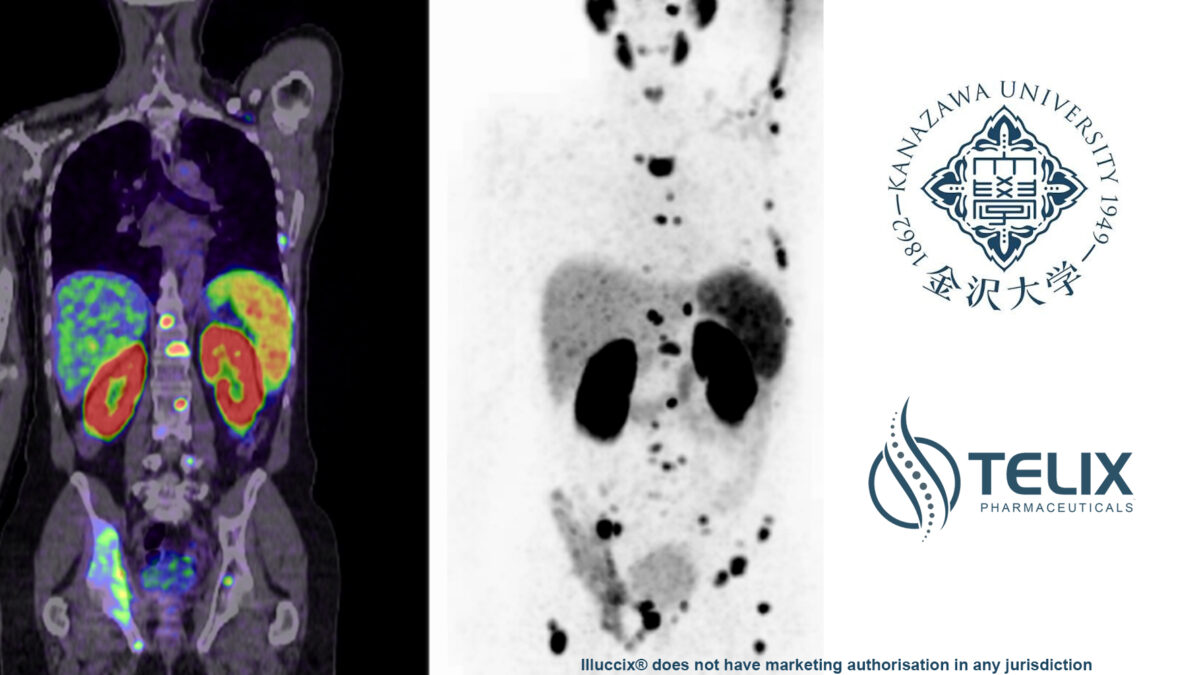
Telix has completed a Phase I clinical trial of TLX591-CDx for prostate cancer imaging in Japan in collaboration with Kanazawa...
Read more
Telix announces collaboration with Lightpoint Medical to explore molecularly targeted radiation and Lightpoint SENSEI® in radioguided surgery...
Read more
Telix announces pan-cancer clinical collaboration with Merck KGaA, Darmstadt,...
Read more
Telix announces two new studies to evaluate the potential utility of TLX591-CDx and TLX250-CDx in breast...
Read more